 |
AboutOpen | 2021; 8: 55-70 ISSN 2465-2628 | DOI: 10.33393/ao.2021.2266 REVIEW |
 |
Overcoming barriers to the use of continuous subcutaneous insulin therapy in type 1 diabetes: a new opportunity from catheter-less pumps
ABSTRACT
Although it represents a valid alternative for patients with type 1 diabetes mellitus (T1DM) with insufficient glycemic control and/or with recurrent hypoglycemia, insulin pump therapy (continuous subcutaneous insulin infusion, CSII) in Italy is nowadays still relatively uncommon. The following review aims to investigate the barriers that limit the transition to CSII and the reasons behind the widespread cases of dropout, particularly evident in younger patients. Among the interventions that could contribute to the solution of the problem, catheter-less insulin pumps (or patch-pumps) will be described: a tool potentially able to reduce, if not eliminate, some of the main obstacles encountered by patients. In particular, a new opportunity has become available today thanks to the evolution of the generation of catheter-less insulin pumps represented by Accu-Chek® Solo (Roche Diabetes Care GmbH); therefore, three clinical cases of patients with T1DM undergoing therapy with the Accu-Chek® Solo insulin pump will be presented and discussed herein.
Keywords: CSII, Glycemic control, Hypoglycemia/prevention and control, Insulin/administration and dosage, Subcutaneous infusion, Type 1 diabetes mellitus
Received: April 19, 2021
Accepted: May 20, 2021
Published online: July 2, 2021
AboutOpen - ISSN 2465-2628 - www.aboutscience.eu/aboutopen
© 2021 The Authors. This article is published by AboutScience and licensed under Creative Commons Attribution-NonCommercial 4.0 International (CC BY-NC 4.0).
Commercial use is not permitted and is subject to Publisher’s permissions. Full information is available at www.aboutscience.eu
Introduction
In subjects with type 1 diabetes mellitus (T1DM), the treatment consists of insulin replacement therapy, a life-saving drug that can never be interrupted (1,2).
Insulin therapy can be administered by multiple daily injections (MDIs) or by continuous subcutaneous insulin infusion (CSII). Numerous evidences have shown that the treatment should be as intensive as possible, that is aimed at achieving the best blood glucose level compensation while minimizing the risk of hypoglycemic episodes, to reduce the risk of complications (1).
When compared with MDI, insulin therapy using CSII allows some improvements in terms of diabetes management in both adult and pediatric patients (3). In fact, the use of the pump allows not only to improve blood glucose level control and decrease episodes of severe hypoglycemia, but also to add benefits in terms of quality of life (QoL) in patients using the pump (4-8).
Both national and international guidelines also recommend, in certain cases, pump therapy in subjects with T1DM. More specifically, the American Diabetes Association along with MDI considers CSII an effective tool to implement intensive diabetes management, having as a goal the achievement of desirable blood glucose levels and improved lifestyle flexibility (9).
At the national level, the AMD-SID Guidelines and the document of the AMD-SID-SIEDP Intercompany Study Group “Technology and Diabetes” recommend evaluating the use of insulin pump for patients with T1DM under certain special conditions, that is, in subjects who, despite intensive and optimized MDI therapy, have glycated hemoglobin (HbA1c) levels persistently above the desirable target and/or have a high risk/episodes of recurrent, severe, or nocturnal hypoglycemia (1,8).
On the other hand, as far as pediatric age subjects are concerned, CSII therapy is recommended in case of: unsatisfactory metabolic compensation (persistently high HbA1c and/or blood glucose level instability), high insulin sensitivity, neonatal or preschool diabetes, recurrent episodes of nocturnal or severe hypoglycemia, or compromised lifestyle with MDI therapy (1,8).
As highlighted in the Guidelines, although it constitutes a valid alternative for patients with insufficient control of blood glucose levels and/or with recurrent hypoglycemia, pump therapy in Italy is still not widespread (10). Patients under CSII actually constitute only 18.1% of the population of interest (in a survey conducted on the national territory, the prevalence of pump use in children and adolescents was found to be 27%) (10,11), a figure that is far from the approximately 40% recorded in the USA, but which is starting to approach almost 20% as recorded in other European countries, such as Norway, Austria, the Netherlands and Switzerland (12).
There are many reasons for said data discrepancy: from the perceived obstacles that limit the transition to pump therapy to, sometimes, certain problems experienced during therapy, which even lead to discontinue pump use.
Understanding the reasons that hinder the start of pump therapy and those that determine its discontinuation is of fundamental importance to be able to identify useful measures to reverse this trend. This review therefore aims to analyze, starting from the data available in the literature today, the barriers and reasons behind this use deadlock, both in adult and younger patients.
Therefore, the thoughts and experiences of adult patients with T1DM, the perceptions of parents of children with T1DM, and the perceptions of healthcare professionals, who play a key role in encouraging and supporting patients throughout the course of therapy, will be examined.
The issues that lead patients to the decision of not wanting to continue traditional pump therapy will be concurrently investigated.
From the findings resulting thereof, among the interventions that could provide a concrete contribution to solving the problem, the proposal to consider catheter-less insulin pumps, the so-called patch-pumps, as a possible tool aimed at reducing, if not eliminating, some of the main obstacles encountered by patients with T1DM will be discussed.
In particular, it will be described how to provide a concrete answer to the specific needs of some patients with T1DM, thanks to the evolution of the catheter-less pump generation, represented by the Accu-Chek® Solo pump (Roche Diabetes Care GmbH).
Small and unobtrusive to carry around, Accu-Chek® Solo understands multiple basal profiles, variable basal rates, and bolus amount settings.
The system ensures high accuracy of delivery, both basal and bolus, and, thanks to the possibility to deliver the bolus directly from the pump, a more comfortable management of the therapy.
Its modular design also allows the pump to be temporarily disconnected, without wasting insulin or plastic components of the instrument.
Three clinical cases of patients with T1DM treated with the Accu-Chek® Solo catheter-less pump will be described to support the benefits arising thereof. The clinical results and the evaluation of individual patients’ perception of the system, obtained through the administration of two questionnaires, will be reported and discussed.
Barriers limiting the transition from MDI to CSII therapy
The experiences of adult patients with T1DM
Barriers to widespread use of a technology can be divided into two main categories: non-modifiable factors and modifiable factors (13).
While the former includes, for example, socioeconomic factors, access to care, and other more strictly demographic factors, the latter includes factors that can be dealt with individually or within the family (13-16). The latter can be investigated, evaluated, and, possibly, resolved with an adequate support of the diabetes team and with the selection of the most appropriate device for the particular characteristics of the patient (13).
Starting from these assumptions, a recent survey proposed to analyze, through the submission of a questionnaire, the main obstacles that would prevent a broader diffusion of devices for diabetes, also including CSII therapy, in a sample of 1,503 adult patients participating in the T1DX Clinic Registry (13).
Having to consider a list containing 19 possible barriers that would limit the use of diabetes technologies, including both non-modifiable and modifiable factors, the participants identified cost-related barriers as the most common barriers. In detail: for 61% the cost of supplies, for 57% the cost of the device, and for 57% the insurance coverage. These reasons are limited to countries with an insurance-based healthcare system and are among the socioeconomic factors that cannot be modified at the level of the individual. Next, barriers related to the experience of wearing the devices, including discomfort with having to wear them (47%) and not liking having diabetes devices on one’s body (35%), were reported as most common.
Taking into account the age of the participants, the two younger groups of patients (18-25 years; 26-34 years) identified more barriers than the other two groups (35-50 years; >50 years), especially in terms of “wearability” of the devices (13).
In addition, the two younger groups expressed more frequently feeling nervous about having to rely on the technology; the younger group of patients (18-25 years old) also showed greater concern about “what others think” (17%) and “not wanting other people to notice or ask questions about their devices” (16.9%), unlike older age groups of patients (3.6%-9%) (13).
The survey also revealed a gender difference in perceptions and barriers to the use of the devices, which were more prevalent in female than male respondents:
– the “annoyance of wearing devices all the time” (51% vs 43%);
– the “not liking the appearance of diabetes devices on their bodies” (30% vs 20%), as well as perceiving a higher level of stress (2.1 vs 1.9), assessed from the Diabetes Distress Scale (13).
The perspective of healthcare professionals
Endocrinologists/diabetes specialists, nurses, dietitians, and other healthcare professionals play a critical role in promoting compliance of insulin therapy devices (17). The diabetes team represents the main resource for the patient in terms of structured education, support and resolution of issues related to the use of the devices and can facilitate the initial transition to the new technology (18). A study assessed healthcare professionals’ opinions on what would be the main issues and barriers in patients’ use of the devices, including CSII therapy. The perception of healthcare professionals was then compared with the survey carried out directly with T1DM patients (17).
A list of 19 possible barrier factors was previously prepared, also starting from the data obtained from the previous patient survey; 209 participants decided to answer the online survey (17).
Healthcare providers identified an average of 7.2 barriers (Standard Deviation [SD] = 3.5). Again, the most commonly cited barriers belong to the category of non-modifiable factors (insurance coverage, costs). Focusing instead on modifiable factors, the barriers most frequently reported by healthcare professionals are:
– the perception that their patients do not like to wear the device (73%);
– that patients do not like the number of pump alarms (40.7%);
– that patients do not understand what to do with the information or with the system features (42%) (17).
Comparing the results of the survey addressed to adult patients with the results of this second survey, healthcare professionals seem to identify the presence of barriers more often than adult patients with T1DM.
In particular, this emerged prominently on the question of not liking to wear the device, with a percentage of 64%-73% compared to only 35% of adults with T1DM. Furthermore, a great discrepancy was found regarding the perception of the patient’s ability to make the most of the information received from the device: almost half of healthcare workers suggested that this was an obstacle for their patients, as opposed to a small percentage among adults with T1DM (17).
Failure to align the perceptions of patients and healthcare professionals could have negative repercussions on the uptake and compliance to diabetes management devices (17).
The case of pediatric patients
Today, insulin pump therapy failure to become widespread poses an issue especially for the pediatric population (3).
In a qualitative study conducted by Sullivan-Bolyai et al, although it was reported that parents of children with T1DM observed better perceptions on glucose level control, as well as easier day-to-day diabetes management, the widespread use of the insulin pump is still low in pediatric patients (3,19).
Based on these considerations, a study set out to analyze, among parents of children aged <7 years and with a duration of illness ≥1 year, what the barriers to insulin pump switching were in the group of parents who, despite receiving a recommendation from their Diabetes Specialist, had decided not to start CSII therapy (3).
Therefore, a survey was conducted on 97 parents of children with diabetes, not in therapy with pump. They were asked to evaluate a list of 13 potential barriers or reasons to explain their refusal. From the analysis of the results, the main barriers reported by parents of pediatric patients were (3):
– the physical burden on the child;
– apprehension about the therapeutic efficacy of the insulin pump.
Therefore, it would seem that the concern related to physical discomfort in wearing the technology outweighs the diabetes specialist’s suggestion to use the devices proven to be effective. The main concerns of discomfort relate, in general, to a sense of physical discomfort, also due to the interference of the cannula on daily life (71%) and on sports activities (59%), the use of infusion sets and catheters (53%) in addition to skin reactions at the insertion sites (51%) and the child discomfort due to wearing a device (46%) and an insulin pump, which could be too big (45%).
For the second barrier identified regarding therapeutic efficacy, the percentage that emerged could be related to being more familiar with and to routine use of MDI therapy. Many patients expressed concerns about hypoglycemia as a consequence of pump use, suggesting that they either lacked adequate information about its potential benefits or held certain beliefs about some of its negative effects. However, since only 12% of patients’ parents reported that they had not received adequate support from their key healthcare professional, doubts about effectiveness may be the result of personal beliefs. For example, some of them may feel uncomfortable with diabetes management technologies and prefer insulin injection, because they are more accustomed to this mode of administration, which is considered more controllable (3).
Drop-out cases in CSII therapy: a widespread phenomenon among adolescents and young adults with T1DM
For a better understanding of the problem, it is also important to evaluate the reasons that lead some subjects to discontinue the use of the pump after an adequate period of testing and treatment. This situation seems to be frequent among children and adolescents with T1DM (in Italy the discontinuation rate is 6.1%), in whom the insulin pump is not always well tolerated or, even less, accepted, despite considerable scientific evidence supporting its use in this group of patients, in order to obtain a better metabolic control (18,20).
Some studies have identified, among the factors associated with cases of drop-out found among adolescents and young adults, a greater degree of glycometabolic decompensation (higher HbA1c levels, more frequent episodes of diabetic ketoacidosis in the last year, reduced self-monitoring of capillary glycaemia), age (the highest percentage of discontinuation would be between 10 and 15 years of age), gender (particularly females), and general well-being (discontinued CSII therapy is greater among patients suffering from depressive symptoms) (21-24).
Common reasons for discontinuation in adolescents include: diabetes burnout (fatigue related to having to change the pump site, monitoring blood glucose levels, and having to apply carbohydrate counting), concerns about body image and potential weight gain, interference with sports and other activities, and a desire to be free of an external device on the patient’s body (18,21,24).
A study conducted in Germany in 2010 was the first to directly ask adolescents and young people about the advantages and disadvantages associated with the use of the insulin pump, starting from a questionnaire developed with input from diabetes patients themselves in an ad hoc focus group (18).
Of the 88 participants, patients who had previously used an insulin pump justified their decision to discontinue therapy with reasons especially related to the catheter and social/psychological factors (e.g., the visibility of the device and the feeling of carrying an external device).
Mainly it was found that the motivations were not dependent on problems of a technical nature of the device; in fact, only 15% of former users identified technical problems concerning the pump, its malfunctioning, or the unpleasant use in public as the determining factors in the decision not to continue with the therapy. Even clinical factors did not appear to be critical in the decision to end CSII therapy (only 22% of patients discontinued therapy for worsening of blood glucose levels) (18).
Finally, when asked to rank a range of advantages and disadvantages in terms of social/psychological factors, former users were less likely to associate an improvement in well-being and independence with pump use. Conversely, they were instead likely to consider the pump as a constant reminder of their diabetes, considering it annoying during daily activities as well as perceiving it as an external device (18).
When gender and age are taken into account, participants showed these differences:
– The boys were less likely to agree that the pump requires more discipline in measuring blood glucose and said that the pump allows better blood glucose regulation throughout the day;
– Young girls showed greater agreement that the pump is more visible and its sound is more annoying;
– Patients >19 years of age considered the pump more annoying during different activities than the younger age group (18).
In conclusion, when asked to identify prerequisites for a possible resuming of CSII therapy, the most common response indicated an improvement of the catheter, followed by a request to refine the management of the pump (size reduction, innovative design, water resistance) and to integrate a blood glucose measuring device into the system (18).
Again considering participants in the T1D Exchange Clinic Network Registry as the starting sample, a more recent study set out to (21):
– Determine the frequency of discontinuation of pump use 1 year after recruitment;
– Identify the factors associated with the decision to discontinue therapy among patients using newer-generation insulin pumps in the United States;
– Summarize the main reasons why patients no longer wanted to continue CSII therapy.
The cohort included in this analysis consisted of 8,935 participants from 67 clinics in the T1DM Exchange Clinic Network. Although patients of all ages were initially included in the cohort, only children, adolescents and young adults were considered for the more in-depth analyses, as the discontinuation rate among patients aged >26 years was not sufficiently relevant (1% vs 2% in the 6-13 years range, 4% for both 13 to <18 years and 18 to <26 years).
Using a list containing 21 reasons, those who, 1 year after enrolment, had decided to discontinue CSII were asked about the reasons related to their decision to stop continuing insulin pump therapy (21).
The reasons associated with discontinuation were relatively similar in the different age groups. Specifically, 57% of participants had device fitting problems (including issues with insertion), discomfort, skin reactions, and problems associated with device adhesion and interference with sports and other activities (21).
Other common reasons included: not liking the pump or feeling anxious (44%), having problems controlling blood glucose levels while on CSII therapy (30%), recommendations from the healthcare provider (20%), not finding the pump helpful (19%), and experiencing problems with device operation (19%).
Above all, girls between 6 and 18 years of age reported problems associated with the fit of the device, a consideration that, however, is also very relevant in boys between 18 and <26 years of age, although the frequency of discontinuation of therapy was lower in this group. Assessing all ages involved in the survey, more girls than boys discontinued CSII therapy because they did not like the device or felt anxious (21).
Beyond the barriers to the use of CSII therapy
Considering the clinical picture outlined so far, it is necessary to develop targeted and personalized interventions that take into account the problems that have emerged, whether related to the transition to CSII therapy or to its interruption, as well as the age and therapeutic experience of the patient.
With respect to barriers and issues associated with device wearability, such as the discomfort of carrying an external device on one’s body, concerns about body image, and discomfort experienced during sports and other activities where the presence of the catheter emerges as a major problem, catheter-less insulin pumps could provide an important aid. To date, several types of insulin pumps are available (8,25).
The models differ in their management, materials used, functions, design and integration with Continuous Glucose Monitoring (CGM) (25).
Recently, smaller, more discreet pumps have entered the market: catheter-less pumps, or patch-pumps, which get their name from the fact that they adhere to the body with an adhesive patch. These devices have been developed to meet the needs and preferences of patients, particularly with regard to their desire to have systems without a visible infusion set (25).
In general, catheter-less insulin pumps are smaller, more discreet, easier to use, and often more cost-effective than traditional insulin pumps.
Given the heterogeneity that characterizes patch-pumps, it is difficult to compare the different models, which differ mainly in size, shape and technology used (26).
Most catheter-less pumps have an additional remote control to control the pump itself; however, some can also be used without an external control system, that is, the insulin bolus can be delivered as needed directly with a button on the pump (26).
Compared to conventional pumps, patch pumps have several advantages, but also some limitations.
As regards the latter, they include, for example (26):
– a waste of insulin when the patch-pump needs to be replaced and the remaining insulin was not used;
– a risk of allergic reactions to the patch;
– a risk of premature detachment of the patch from the site of administration;
– a waste of plastic/metal material;
– the infusion site is not visible.
On the other hand, as far as benefits are concerned, they include (26):
– smaller size and lighter devices than traditional pumps;
– they do not require a catheter, they pose fewer problems regarding the insulin infusion set (IIS) (catheter/needle/tube), and their needle is not visible;
– the IIS system is inside the pump, or in the base of the patch-pump;
– reduced risk of insulin obstruction in the IIS system, as insulin remains at a stable temperature level within the patch-pump;
– full freedom of movement;
– ease of use, easier handling, design features appreciated by patients;
– simplified training, fewer steps to start CSII therapy;
– the pump may be placed on different parts of the body to ensure greater discretion and proper rotation of application sites;
– the application of the device on the body is almost painless;
– many patch-pumps allow remote control of the insulin infusion rate (like several traditional mini-dosers);
– in some patch-pumps, bolus calculators are integrated in the remote control device.
The set of specific features described so far make catheter-less pumps a new opportunity to address the many problems experienced by patients with T1DM, both at the time of transition to CSII therapy and to maintain therapeutic compliance.
As with all new generations of technology, certain cautions and cares are required (25):
– being able to understand different basal profiles, varying basal rates, and insulin bolus amount settings;
– ensuring basal and bolus delivery with high accuracy;
– managing therapy more conveniently through the ability to deliver boluses directly from the pump, independent of the diabetes management tool;
– a modular design that allows the pump to be temporarily disconnected without having to replace the entire pod, thus avoiding unnecessary waste of insulin or plastic pump parts.
Therefore, the need arose to develop a catheter-less insulin pump that would not only be small and discreet to carry around, but would also meet these specific needs. The answer has come from the evolution of the catheter-less pump generation: Accu-Chek® Solo (Roche Diabetes Care GmbH), consisting of a pump without catheter with remote control (Fig. 1) (25).

Fig. 1 - Diabetes manager and pump.
Patch-pump and MDI compared: the state of the art
Although several studies have investigated and demonstrated the benefits of CSII therapy in patients with T1DM as compared to MDI, the same cannot yet be said for patch-pumps (9,26), due to the limited amount of scientific evidence.
From a literature search conducted on PubMed, it is immediately apparent that the number of clinical studies in which this issue has been appropriately addressed is very small. If a multicenter, retrospective study proposed to evaluate the efficacy of a patch-pump in patients with type 2 diabetes mellitus (T2DM) in comparison with MDI therapy (27), to date, there are three studies that have considered this comparison in patients with T1DM (28-30).
Specifically, in the retrospective study conducted by Danne et al (29), from the German/Austrian Diabetes Patienten Verlaufsdokumentation registry, data from 2,529 patients with T1DM, referred to 263 centers, aged <20 years, were analyzed to evaluate blood glucose level control in patients who had switched from MDI to CSII using a patch-pump, compared with those who had maintained MDI. The results obtained showed that, at 1 year after the switch, blood glucose levels control was better in the first group of patients (7.5% ± 0.03% vs 7.7% ± 0.02%), p < 0.001; however, no differences were recorded at 2 and 3 years. The daily insulin dose was also lower in the patch-pump therapy group (p < 0.001): 0.80 ± 0.01, 0.81 ± 0.01 and 0.85 ± 0.01 U/kg vs 0.89 ± 0.01, 0.94 ± 0.01 and 0.97 ± 0.01 U/kg (at 1, 2 and 3 years, respectively, p < 0.001), while the Body Mass Index (BMI) Standard Deviation Score (SDS) value increased in both groups considered. Recent results are presented in the multicenter, retrospective study conducted by Mehta et al on 156 patients with T1DM who, coming from MDI therapy or CSII (with catheter pump), started using a patch-pump (30). Use of the catheter-less device for 12 months was associated with a significant overall improvement (considering both patient groups) in A1c, amounting to a reduction of −0.3% (95% confidence interval [CI] from −0.5 to −0.1%, p = 0.007), or from 8.1% ± 1.5% to 7.8% ± 1.3%. Specifically, the group of patients who switched from MDI therapy (n = 99) to patch-pump therapy had a significant decrease in A1c of −0.4% (from 8.2% ± 1.6% to 7.8% ± 1.2%, p = 0.009). Although the mean variation was greater for these patients, the difference between the two groups examined was not significant (p = 0.1). Finally, improvements in A1c values were obtained without any body weight gain in the patients (30).
Given the clinical findings, appropriate clinical trials shall be planned and will be necessary in the near future, in order to compare parallel groups in therapy with traditional CSII, catheter-less patch-pump, and insulin pens. The purpose of these studies should be to investigate clinically relevant endpoints, considering adequate samples in terms of sample size and representativeness. From all of this, specific economic analyses could be initiated in addition to patient-reported outcomes (PROs). PROs are actually an essential tool to understand both the burden and the benefits of the devices under investigation, allowing, at the same time, to evaluate their impact on the daily life of patients (26,30).
In this regard, the first 6-month results of the PRO Solo clinical study (Clinical Trial Registration Number: NCT03478969) have just recently been presented during the 56th Congress of the European Association for the Study of Diabetes (EASD) (31).
In this multinational, multicenter, controlled, randomized, three-arm study, 181 patients with T1DM (16 of whom subsequently withdrew for various reasons), naïve to CSII therapy, were enrolled with the objective of analyzing the effect of the Accu-Chek® Solo pump on PROs. Participants used the Accu-Chek® Solo (ACS) pump (n = 55), MDI therapy (n = 58) or the Insulet Omnipod® (IO) pump (n = 52) for 6 months and then continued with the ACS pump for an additional 3 months. The primary endpoint was the Diabetes Technology Questionnaire (DTQ), administered at baseline and at 3, 6, and 9 months, and the secondary endpoints were HbA1c values and the Problem Areas in Diabetes (PAID-5) (31).
Regarding the 6-month results, the mean change for DTQ scores was respectively: 105.1 ± 20.2, 94.9 ± 11.8, and 107.5 ± 17.4 (higher scores reflect more positive PROs). Hierarchical tests reported a significant increase in PROs among ACS users compared with those on MDI therapy (104.4 vs 94.5 ANCOVA, p < 0.01), whereas no difference emerged between patients using ACS and those on IO (104.7 vs 108.7, p = 0.33). Similarly, HbA1c values improved significantly in the group of patients receiving ACS therapy compared to the group on MDI (−0.10% +0.25%, p = <0.01), a difference that did not emerge in the case, instead of patients receiving IO therapy (−0.04% vs −0.02%, p = 0.90) (31).
The DTQ results showed a significant improvement, for all parameters considered, in patients using the pump compared to patients on MDI. Specifically, confidence about taking a correct dose of insulin during exercise, on sick days, or when meals are skipped or delayed was high; while visibility of the disease and interference in daily life were low. In addition, concerns about long-term health and the fear of experiencing hypoglycemia at night have decreased. Finally, PAID-5 scores also showed a significant improvement in patients on ACS therapy compared to those on MDI therapy (6.32 vs 7.62, p = 0.01), but not in the case of the comparison with the group that used the IO pump (6.00 vs 6.05, p = 0.93) (31).
In conclusion, patients who used the Accu-Chek® Solo pump had a significantly positive impact both in terms of consistent improvement in all psychosocial factors investigated and in terms of HbA1c. From these results, it can be deduced that the Accu-Chek® Solo pump can be considered a valid therapeutic choice for patients with T1DM (31).
Three clinical cases in comparison
Below are three clinical cases of patients with T1DM treated with Accu-Chek® Solo, two adult patients and one pediatric patient respectively.
Clinical case No. 1
The patient, a 32-year-old woman with T1DM since 2011 (Tab. I), has a positive family history of acute myocardial infarction (AMI) (paternal grandfather), leukemia (paternal grandmother), and neoplasm of the head (maternal grandfather). Her job requires her to travel frequently and she regularly attends various social events. She follows a varied diet, does not smoke, and denies the intake of alcoholic beverages. At the recent pathological history, the patient reports to be on basal-bolus insulin therapy and to use Flash Glucose Monitoring (FGM) with alarm application, and performs corrections with additional injections of ultrafast insulin based on values or meal-related needs.
| Patient 1 | Patient 2 | Patient 3 | |
|---|---|---|---|
| Age (years) | 32 | 23 | 7 |
| Gender | W | W | W |
| Duration of diabetes (years) | 9 | 13 | 3 |
| BMI (Kg/m2) | 24.3 | NA | NA |
| Smoker | No | NA | No |
| Diabetes complications | No | No | No |
| Treatment in previous months | |||
| MDI | |||
| MDI + CGM | X | ||
| MDI + FGM | X | X | |
| CSII | |||
| SAP combined | |||
| SAP integrated | |||
BMI = body mass index; CGM = continuous glucose monitoring; CSII = continuous subcutaneous insulin infusion; FGM = flash glucose monitoring; MDI = multiple daily injection; NA = not available; SAP = sensor-augmented pump; T1DM = type 1 diabetes mellitus; W = woman.
At the first evaluation in January 2020, the patient weighed 67.0 kg (BMI 24.3 kg/m²); blood pressure was 115/70 mmHg. Cardio-thoracic-abdominal objectivity is within limits.
With regard to the evaluation of metabolic parameters, fasting blood glucose was 200 mg/dL and HbA1c was 7.4%; low-density lipoprotein (LDL) cholesterol was 105 mg/dL. Renal and hepatic function, electrolytes, and thyroid-stimulating hormone (TSH) are normal. Absence of microalbuminuria was detected. Drug therapy consists of insulin Degludec 14 IU in the evening, insulin faster aspart based on carbohydrate count with insulin-to-carbohydrate ratio of 1/20 at breakfast and lunch and 1/15 at dinner (insulin sensitivity factor 70).
It is suggested that the patient consider switching from MDI therapy to CSII + CGM therapy. Considering her request for a device that is easy to carry and use, the Accu-Chek® pump is placed only after positive feedback and evaluation of clinical data (Tab. II).
At the scheduled checkup after 4 months, we found (Tab. III) an improvement in blood glucose levels compensation (HbA1c from 7.6% to 6.9%), blood glucose levels variability (mean blood glucose levels ranging from 178 to 152 mg/dL) and time percentages spent in the different glycemic ranges. Specifically, for euglycemic range time of 70-180 mg/dL (time in range [TIR]), the increase was from 46% to 70%; hyperglycemia time (time above range [TAR] > 180 mg/dL) decreased from 47% to 28%; finally, hypoglycemia time (time below range [TBR] <70 mg/dL) decreased from 7% to 2%. Among the parameters analyzed, in addition, an increase in the use of CGM from 79% to 100% was observed. The use of CSII allowed the patient to achieve optimization of blood glucose levels control and to increase therapy compliance.
| Baseline Examination – Accu-Chek ® Solo Placement | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| HbA1c (%) | 7.6 | 7.0 | 7.7 |
| Mean blood glucose level (mg/dL) | 178.0 | 156.0 | 173.0 |
| Hypoglycemia episodes (n) | NA | 22 | 0 |
| TIR 70-180 mg/dL (%) | 46.0 | 55.0 | 58.0 |
| TAR >180 mg/dL (%) | 47.0 | 21.0 | 42.0 |
| TBR <70 mg/dL (%) | 7.0 | 4.0 | 0.0 |
| SD (mg/dL) | NA | 82.0 | 65.0 |
| CV (%) | 42.4 | 53.2 | 59 |
| CGM usage time (%) | 79 | 97 | 100 |
CGM = continuous glucose monitoring; CSII = continuous subcutaneous insulin infusion; CV = coefficient of variation; HbA1c = glycated hemoglobin; NA = not available; SD = standard deviation; T1DM = type 1 diabetes mellitus; TAR = time above range; TBR = time below range; TIR = time in range.
| Follow-up | Patient 1 (120 days) | Patient 2 (60 days) | Patient 3 (180 days) |
|---|---|---|---|
| HbA1c (%) | 6.9 | 6.2 | 7.0 |
| Mean blood glucose level (mg/dL) | 152.0 | 131 | 155.0 |
| Hypoglycemia episodes (n) | NA | NA | 0 |
| TIR 70-180 mg/dL (%) | 70 | 83.9 | 70.0 |
| TAR >180 mg/dL (%) | 28.0 | 11.8 | 30.0 |
| TBR <70 mg/dL (%) | 2.0 | 4.3 | 0.0 |
| SD (mg/dL) | NA | 45.5 | 59.0 |
| CV (%) | 57.0 | 34.0 | 35.0 |
| CGM usage time (%) | 100.0 | 100.0 | 100.0 |
CGM = continuous glucose monitoring; CSII = continuous subcutaneous insulin infusion; CV = coefficient of variation; HbA1c = glycated hemoglobin; NA = not available; SD = standard deviation; T1DM = type 1 diabetes mellitus; TAR = time above range; TBR = time below range; TIR = time in range.
At the follow-up visit, in addition to performing the clinical evaluation, the patient is provided a questionnaire on the Continuous Subcutaneous Insulin Infusion-Related Quality of Life (CSII-QoL) scale, to further investigate the impact of CSII on the QoL (Tab. IV) (32).
The 28 questions of the questionnaire must be answered with a value between 0 and 4 (0 = strongly disagree; 1 = disagree; 2 = neither agree or disagree; 3 = agree; 4 = strongly agree). From the analysis of the prevailing factors (Tab. IV), regarding Factor 1 advantages/benefits, the patient “agrees” that insulin pump therapy is useful. On the contrary, it emerges that they “disagree” that pump therapy involves a series of social restrictions (Factor 2), while they “neither agree or disagree” on the impact of the pump on the psychological sphere (Factor 3).
| 0 = strongly disagree
1 = disagree 2 = neither agree or disagree 3 = agree 4 = strongly agree |
|||
| Patient 1 | Patient 2 | Patient 3 | |
|
1. Pump therapy is useful for controlling blood glucose levels, in general |
3 | 4 | 3 |
|
2. Pump therapy is useful to prevent hypoglycemia |
3 | 4 | 3 |
|
3. Pump therapy is useful to correct hyperglycemia |
4 | 4 | 4 |
|
4. It’s easy to change your baseline using pump therapy |
4 | 4 | 4 |
|
5. It is easy to change the amount of insulin delivered to the meal/snack bolus using pump therapy |
4 | 4 | 3 |
|
6. The pump therapy is a relevant financial commitment (given the non-applicability to the Italian socioeconomic reality, this answer may be omitted) |
3 | 3 | |
|
7. Pump therapy is a barrier for working people, professionals, or in a school setting |
0 | 2 | 1 |
|
8. Pump therapy limits the ability to go outdoors |
0 | 2 | 1 |
|
9. Pump therapy limits relationships or social activities |
1 | 2 | 1 |
|
10. Pump therapy limits leisure activities |
1 | 1 | 1 |
|
11. Pump therapy limits clothing choices |
3 | 2 | 3 |
|
12. Pump therapy limits the ability to exercise |
2 | 1 | 1 |
|
13. Pump therapy is difficult to use |
1 | 1 | 1 |
|
14. The pump (or controller) screen is hard to see |
1 | 3 | 1 |
|
15. The pump alarms are disrupting |
2 | 1 | 3 |
|
16. Solving a pump problem takes a lot of effort |
2 | 1 | 2 |
|
17. Replacing the infusion set (or the cannula alone) of the pump requires a lot of effort |
1 | 1 | 1 |
|
18. Wearing the pump is uncomfortable |
2 | 2 | 2 |
|
19. Wearing the pump is a psychological burden |
2 | 2 | 1 |
|
20. Wearing the pump is annoying because it catches people’s attention |
3 | 3 | 2 |
|
21. Skin irritation from the patch is uncomfortable |
2 | 2 | 0 |
|
22. The pain of inserting the cannula is intolerable |
1 | 2 | 1 |
|
23. Finding an infusion site for placing the cannula requires a major effort |
2 | 1 | 1 |
|
24. I’m afraid of hyperglycemia |
1 | 4 | 3 |
|
25. During the day, I’m afraid of hypoglycemia |
3 | 3 | 3 |
|
26. I’m afraid of nocturnal hypoglycemia |
4 | 4 | 4 |
|
27. I am afraid of hypoglycemia when driving a motor vehicle (car, motorcycle) |
3 | 4 | 4 |
|
28. In general, I am satisfied with pump therapy |
3 | 4 | 3 |
| Prevailing factors | Patient 1 | Patient 2 | Patient 3 |
| Factor 1 | 3.5 | 4.0 | 3.3 |
| Advantages/Benefits
(questions no.: 1-2-3-4-5-28) |
|||
| Factor 2 | 1.2 | 2.1 | 1.2 |
| Social restrictions
(questions no: 7-8-9-10-12-13-14-16-20) |
|||
| Factor 3 | 2.1 | 1.7 | 2 |
| Psychological impact
(questions no.: 17-18-19-21-22-23-24-25-26-27) |
|||
Questions 6, 11, and 15 have been excluded from the following analysis.
The patient also agreed to fill in a second questionnaire, developed ad hoc to understand the level of satisfaction of the users of the pump, with a focus on the evaluation of its peculiarities (Tab. V).
Based on the results, the patient is satisfied with Accu-Chek® Solo. In particular, she feels “very satisfied” with regard to the following features: absence of catheter, possibility to deliver bolus directly from the pump and open bolus calculator. Characteristics are considered as “important/extremely important” by the patient. Regarding the simplicity of the pump placement/use procedure, patient found it “very easy” to insert the cannula, deliver the bolus directly from the pump, and deliver the bolus using the diabetes manager. She was “very much in agreement” that the ability to replace components individually provides a high degree of flexibility, that the ability to deliver a bolus directly from the pump provides a feeling of security, that the ability to temporarily remove the pump base makes everyday life easier, and that the pump is simple to use. In particular, patient appreciated the “convenience of the temporal variation of the bolus” the most. When asked if “patient has the intention to continue using the system,” the feedback is positive.
|
1. On the following scale, rate your level of satisfaction with each of the characteristics listed below: 1 = very dissatisfied 2 = dissatisfied 3 = neither dissatisfied nor satisfied 4 = satisfied 5 = very satisfied |
|||
| Patient 1 | Patient 2 | Patient 3 | |
| No catheter | 5 | 5 | 5 |
| Pump removability | 3 | 4 | 4 |
| Bolus may be delivered directly from the pump | 5 | 5 | 5 |
| Reservoir visibility | 4 | 4 | 4 |
| Open bolus calculator | 5 | 3 | 4 |
| Customization of alerts/alarms | 4 | 4 | 4 |
| Pump management via Diabetes Manager (remote control) | 3 | 4 | 5 |
| Ability to view reports directly on the Diabetes Manager (remote control) | 4 | 4 | 5 |
| Video tutorials available directly on Diabetes Manager (remote control) | 4 | 4 | 4 |
| Practical insertion device | 4 | 4 | 5 |
| Device discretion | 4 | 5 | |
|
2. On the following scale, please indicate the level of importance that each of the characteristics listed below has for you: 1 = extremely irrelevant 2 = irrelevant 3 = neither important nor unimportant 4 = important 5 = extremely important |
|||
| Patient 1 | Patient 2 | Patient 3 | |
| No catheter | 4 | 5 | 5 |
| Pump removability | 5 | 5 | 4 |
| Bolus may be delivered directly from the pump | 5 | 5 | 5 |
| Reservoir visibility | 5 | 4 | 4 |
| Open bolus calculator | 5 | 4 | 5 |
| Customization of alerts/alarms | 3 | 4 | 4 |
| Pump management via Diabetes Manager (remote control) | 4 | 4 | 4 |
| Ability to view reports directly on the Diabetes Manager (remote control) | 4 | 4 | 4 |
| Video tutorials available directly on the Diabetes Manager (remote control) | 2 | 5 | 4 |
| Practical insertion device | 5 | 5 | 4 |
| Device discretion | 5 | 5 | 5 |
|
3. On the following scale, rate the ease of placement/use of the Accu-Chek® Solo pump: 1 = very difficult 2 = difficult 3 = neither difficult nor easy 4 = easy 5 = very easy |
|||
| Patient 1 | Patient 2 | Patient 3 | |
| Filling the reservoir | 3 | 5 | 4 |
| Inserting the cannula | 5 | 5 | 4 |
| Positioning the base on the stand | 4 | 4 | 4 |
| Bolus delivery directly from the pump | 5 | 4 | 4 |
| Bolus delivery using the Diabetes Manager (remote control) | 5 | 5 | 4 |
|
4. How much do you agree with the following statements? 1 = very much disagree 2 = disagree 3 = neither disagree nor agree 4 = agree 5 = very much agree |
|||
| Patient 1 | Patient 2 | Patient 3 | |
| The ability to replace individual components gives me a high degree of flexibility | 5 | 5 | 4 |
| The possibility of delivering a bolus directly from the pump gives me confidence | 5 | 5 | 5 |
| The possibility of temporarily removing the base of the pump makes my daily activities easier | 5 | 4 | 4 |
| The size of the micro-pump guarantees me maximum discretion | 3 | 4 | 4 |
| The pump is easy to use | 5 | 5 | 4 |
| The ease of use of the system allows me to think less about diabetes | 4 | 3 | 4 |
| I have experienced significant issues using the pump | 4 | 2 | |
| I had technical problems with the pump | 4 | 2 | |
| I had problems with air bubbles while using the pump | 5 | 2 | |
| If I had any problems, the support service helped me solve them | 5 | 5 | |
| I had to go to the ER for glycemic issues related to using the pump | 1 | 2 | |
|
5. During this first period of use, how satisfied are you with the system? 1 = very dissatisfied 2 = dissatisfied 3 = neither dissatisfied nor satisfied 4 = satisfied 5 = very satisfied |
|||
| Patient 1 | Patient 2 | Patient 3 | |
| 4 | 5 | ||
|
6. Please state what you value most about using the Accu-Chek® Solo pump (open-ended question). Patient 1: Convenience of the temporal variation of the bolus Patient 2: The fact that it has no catheter, it is small and discreet Patient 3 |
|||
|
7. Please state what you least appreciate about using the Accu-Chek® Solo pump (open-ended question). Patient 1: Air bubbles sometimes do not allow correct dose release Patient 2: Not connected to a sensor in an integrated system Patient 3 |
|||
|
8. Is this the first pump you’ve used? Y/N Patient 1: Y Patient 2 Patient 3: Y |
|||
|
9. If, on the contrary, you were already in therapy with a pump, compared to the previous instrument, what is the level of satisfaction with the new model? 1 = very dissatisfied 2 = dissatisfied 3 = neither dissatisfied nor satisfied 4 = satisfied 5 = very satisfied |
|||
|
10. Would you recommend friends, family, and/or acquaintances to use the system? Y/N Patient 1 Patient 2: Y. I gained the courage to make my own decisions and felt I could manage my diabetes (even in quarantine) Patient 3: Y |
|||
|
11. Are you willing to continue using the system? Y/N Patient 1: Y Patient 2: Y Patient 3: Y |
|||
|
12. If not, why? |
|||
In conclusion, the patient is satisfied with her pump therapy and, specifically, with the Accu-Chek® Solo device, because it met her needs and allowed her to better manage unexpected events.
Clinical case No. 2
The patient is a 23-year-old woman with T1DM since 2007 (Tab. I), has no family history of endocrine/metabolic disorders. A college student, she has an active lifestyle and healthy eating habits. There is nothing relevant to report in the physiological history and the patient does not report any recent relevant pathologies. From 2014 to 2018 she is using CSII therapy (MiniMed Paradigm®Veo™), currently on MDI therapy and intermittent blood glucose monitoring (FreeStyle Libre).
At the first evaluation at the Diabetes Care and Management Technology Outpatient Clinic in November 2019, the patient appears to be in good health. General physical examination is normal. Regarding the evaluation of metabolic parameters, blood glucose was 189 mg/dL and HbA1c was 7.5%. Creatinine is 0.8 mg/dL; free triiodothyroinine (FT3) is 3.41 pg/mL, free throxine (FT4) is 1.24 ng/dL, and TSH 2.98 μU/mL. As for the blood count, there is nothing to report. Finally, after complete urine analysis, glucose was 100 mg/dL. Drug therapy consists of insulin Degludec 23 IU in the morning and insulin Lispro 5-6 IU at breakfast, 6-7 IU at lunch and 5-6 IU at dinner.
Considering the clinical picture of the patient and following the question whether today it would be possible to have smaller, less bulky, more discreet, and, above all, catheter-free devices, the feasibility of switching again to CSII is discussed. The patient decides to begin treatment with Accu-Chek® Solo, a device that addresses the need to avoid using a catheter-based pump, a major reason for her previous withdrawal from CSII therapy, and that can better accommodate her active lifestyle. In addition, through carbohydrate counting (CHO counting), the patient can adjust insulin boluses, at meals and beyond, and act in time, correcting glycemic values (through CHO counting and insulin) without having to worry about where she is. Therefore, we proceed to the positioning of the device after evaluating the clinical data (Tab. II).
At the control visit after 2 months, we observe (Tab. III) an improvement in blood glucose levels compensation (HbA1c from 7.0% to 6.2%) and blood glucose levels variability (mean blood glucose levels from 156 mg/dL to 131 mg/dL; coefficient of variation [CV] from 53.2% to 34%). TIR 70-180 mg/dL increased from 55% at baseline to 83.9% at follow-up; TAR >180 mg/dL decreased from 21% to 11.8%, while TBR <70 mg/dL was essentially unchanged (4% vs 4.3%). The CGM utilization rate from 97% reaches 100%.
At follow-up, the following questionnaire is also provided to the patient: CSII-QoL scale (Tab. IV) (32). From the analysis of the prevailing factors (Tab. IV), the patient “strongly agrees” that pump therapy has a number of advantages/benefits (Factor 1); she “neither agrees or disagrees” on Factor 2 regarding social restrictions, while she “disagrees” that the pump has a psychological impact on her (Factor 3).
The patient also decided to fill out the second questionnaire on user satisfaction (Tab. V). Analysis of the responses reported a high level of satisfaction with the Accu-Chek® Solo pump. In detail, the patient was “very satisfied” with the absence of the catheter and the possibility to deliver the bolus directly from the device, factors she considered “extremely important.” “Very easy” is the rating reserved for the simplicity of the procedure of filling the reservoir, inserting the cannula and delivering the bolus, using the Diabetes Manager. The patient “very much agrees” that the ability to replace components individually provides a high degree of flexibility, that the ability to deliver a bolus directly from the pump provides safety, and that the device is easy to use.
Accu-Chek® Solo is especially valued for its absence of a catheter, small size, and discreetness. In addition, the patient would recommend to friends, relatives, and/or acquaintances the use of the system, emphasizing how she had “taken the courage to make decisions independently” and felt “that she could manage her diabetes even in quarantine.” When asked “whether she is willing to continue using the system,” the answer is positive.
In general, the patient is satisfied with pump therapy (“strongly agree”) and, in particular, “very satisfied” with the Accu-Chek® Solo pump. Its use has allowed her to rely on a more convenient and flexible therapy.
Clinical case No. 3
The patient is a 7-year-old girl with T1DM since 2017 (Tab. I). Physical and psychological development was normal. The remote pathological history reports recurrent asthmatic bronchitis, while, at the recent history, the parents report accessing the emergency room (ER) for diabetic ketoacidosis in July 2017. The patient is on insulin therapy and uses CGM (Dexcom G6).
At the time of presentation in October 2019, the physical examination shows a good overall condition.
In order to achieve a better glycometabolic profile, to control the “dawn effect,” and to meet the child’s wish of “not having wires on her body,” the possibility of using CSII therapy with Accu-Chek® Solo pump is proposed. Following the positive feedback from parents and the evaluation of clinical data (Tab. II), the catheter-less pump is placed.
After 6 months of use, at the follow-up visit, there was an improvement in blood glucose levels control (HbA1c from 7.7% to 7.0%) and in blood glucose levels variability (mean blood glucose levels from 173 mg/dL to 155 mg/dL; CV from 59% to 35%). TIR 70-180 mg/dL increased from 58% to 70%; TBR >180 mg/dL increased from 42% to 30%; TBR <70 mg/dL remained unchanged. The CGM use rate is maintained at 100%.
The patient was able to benefit from the transition to CSII therapy, particularly with regard to the goal of improving nocturnal hyperglycemia.
At the follow-up, the questionnaire CSII-QoL scale is provided to the parents of the child (Tab. IV) (32). From the analysis of the prevailing factors (Tab. IV), it emerges that parents “agree” that pump therapy brings a series of advantages/benefits (Factor 1); they “disagree” on Factor 2 (i.e., that pump therapy brings a series of social restrictions), while they “neither agree or disagree” on the fact that the pump has a psychological impact (Factor 3). The patient’s parents also consented to complete the second questionnaire (Tab. V). When asked to evaluate the level of satisfaction regarding the characteristics of the pump, a high level of satisfaction (“very satisfied”) was found regarding the absence of a catheter, the possibility of delivering bolus directly from the pump, management of the pump by the Diabetes Manager, the possibility of viewing reports directly on the Diabetes Manager, practicality of the insertion device, and discretion. These characteristics were considered “important/extremely important” by the patient’s parents.
“Easy” is considered all Accu-Chek® Solo placement/use procedures: filling the reservoir, inserting the cannula, placing the base on the stand, delivering the bolus directly from the pump, and delivering the bolus using the Diabetes Manager.
They considered themselves to be “very much in agreement” that the ability to deliver a bolus directly from the pump provides safety and generally “in agreement” with other observations, such as the fact that the size of the pump provides maximum discretion, that the ease of use of the system allows them to think less about diabetes, and that the ability to temporarily remove the base of the pump facilitates daily living.
When asked if “you would recommend the use of the pump to friends, relatives, and/or acquaintances” and if “you intend to continue using the system,” the response is always positive.
In general, attentive and supportive parents are satisfied with the technology, especially with the ability to deliver a bolus in any condition, even directly from the device. The impact on daily life is positive, because the Accu-Chek® Solo pump has made it possible to almost eliminate the “social discomfort” of having to be away from people to perform a correction.
Discussion
Despite being able to more accurately reproduce physiological insulin secretion and making more accurate and precise dose adjustments possible, bringing potential benefits to patients, the widespread CSII therapy in Italy is still too limited (8,10).
Therefore, the purpose of this review is to investigate, through careful analysis of the most recent literature, on the one hand, the barriers that would limit the transition to CSII therapy, and, on the other hand, the reasons that would induce some patients with T1DM to abandon the pump therapy.
Regarding the first question, the most common barriers were found to be those associated with cost, a finding shared by both adult patients with T1DM and healthcare professionals themselves (13,17).
Regarding this result, a clarification is necessary: the studies taken into account refer to the health context of the USA. It is well known, however, that there are considerable differences in terms of CSII costs between countries (26).
In Italy, for example, insulin pumps (both standard models and patch-pumps) are reimbursed by the National Health Service (NHS) in all regions (33).
In T1DM and in the case of diabetes during pregnancy, it is always possible to be prescribed by the National Health Service. The Ministry of Health’s Circular Letter of April 19, 1988, to which the regional resolutions refer, provides a series of requirements for Diabetes Services that indicate pump therapy. They should put in place “direct supervision,” taking charge of patients through intensive supervision criteria and ensuring a 24-hour consultative relationship. These requirements are completed in the regional deliberations, providing that the Centre is distinguished by proven experience in CSII and by the task of educating the patient from the technical and therapeutic point of view, in addition to verifying the achievement of therapeutic objectives (33).
One thing to bear in mind, however, is what emerged from a literature review, in which 11 studies were examined on the cost-effectiveness of CSII compared with MDI. In patients with T1DM characterized by poor metabolic control and/or frequent and/or problematic hypoglycemia, CSII would be cost-effective, due to the decrease in HbA1c values and the lower frequency of hypoglycemia episodes (8,34).
For example, in the case of the survey aimed at assessing the perspective of healthcare professionals, among the most frequently encountered barriers was the indication “that patients do not understand what to do with the information or system features” with 42% (17).
In this regard, in the document of the inter-society study group AMD-SID-SIEDP “Technology and Diabetes,” among the characteristics of the “ideal patient” it is reported that he/she should “be able to learn the use and management of the pump.” Obviously, in the case of the “ideal pediatric patient,” this feature reflects on the role of the family (or of the reference adult), called to ensure an adequate technical and practical support to the management of the pump therapy (8).
It is therefore crucial that the right amount of learning and technical training is provided so that the patient or family member is able to make full use of the device and all of its advanced features.
At the same time, the possibility of providing an appropriate learning pathway could help to dispel the prejudices against CSII therapy found in some parents of children with T1DM. Knowing how to recognize possible resistance behaviors, possibly identifiable through standardized methods, could help healthcare professionals to define the best strategy to undertake (17), as well as an intervention planning that can include moments of technical training in which, thanks to in-depth learning and evaluation of the potential benefits derived from CSII, possible prejudices can be eliminated (8).
In all this, it should also be kept in mind the need for periodic updating checks and moments of educational reinforcement, with the aim of promoting the use of the device and, if necessary, of being able to intervene early if problems emerge (8,17).
Proper attention should also be paid to the psychological sphere, which is not a secondary aspect. In fact, it is important that the patient is motivated, wishes to achieve and maintain a good management of the disease over time, demonstrates emotional stability in the therapeutic pathway, accepts the instrument, and has realistic expectations toward the CSII therapy (8).
The possibility of also developing cadenced support pathways, which help patients (or caregivers) to cope with the different phases of therapy, could actively contribute both to reducing barriers to transition to CSII and to decreasing cases of therapy drop-outs.
Certainly, the barriers associated with the wearability, such as the discomfort of having to carry an external device and the worries about how their body could appear, in addition to the discomfort encountered during sports and other activities (in which the presence of the catheter is one of the main problems), were very frequently reported by the people interviewed in the different surveys (3,13,17,18,21).
One answer to these problems has come from catheter-less insulin pumps, the so-called patch-pumps.
Compared with traditional insulin pumps, patch pumps, which are smaller in size and have no catheter, have been designed to make the infusion set invisible and, possibly, contribute to increase therapy compliance, relying on a discreet system (25). Among the different models available today, a catheter-less pump has been recently introduced on the market. Thanks to its modularity and flexibility, its remote control and the possibility of bolus administration directly from the pump, it has been able to encompass the advantages of both traditional insulin pumps and catheter-less pumps. This is Accu-Chek® Solo (Roche Diabetes Care GmbH), a pump without a catheter (Fig. 1) (25).
The insulin delivery accuracy of the Accu-Chek® Solo (ACS) pump was recently evaluated in comparison to the A6 TouchCare® System (A6) and Omnipod® (OP) patch-pumps, according to the international standard IEC 60601-2-24. For most of the parameters tested, the Accu-Chek® Solo device was more accurate than the A6 pump compared to the OP pump. Specifically, regarding the delivery of the 3 boluses of different volume (0.2 U, 1 U and 10 U), the accuracy of ACS was higher in the case of the individual boluses of 0.2 U and 1.0 U (percentage of individual boluses within ±15% from the target: ACS = 88% and 99%, respectively, vs A6 = 40% and 65%, respectively, and vs OP = 57% and 77%, respectively), whereas in the case of the 10 U bolus, no difference was found between the different models tested. Regarding the accuracy of baseline delivery flow rate, ACS in particular demonstrated significantly more accurate delivery at the rate of 1 U/h (1 h window percentage within ±15% of the target: ACS = 98% vs A6 = 60% and vs OP = 81%). The results between the two models ACS and A6 were similar in the case of the 0.1 U/h rate (ACS = 51% vs A6 = 48%); however, it was higher than those of the OP device (32%) (35).
The Accu-Chek® Solo pump (Fig. 2) consists of three main components (pump base, reservoir, and pump stand), which provide patients with significant system flexibility and modularity. The base of the pump adheres to the body with an adhesive patch and allows the cannula to be secured in place (available in two lengths: 6 mm or 9 mm). This allows the patient to easily insert or detach the pump (25).
The insertion system has been designed so that it can be operated simply with one hand, making independent insertion possible. Moreover, taking into account its small size, the pump can be worn in different parts of the body: in the abdominal region, on the upper arm and on the outer thighs, as well as on the hips (25).
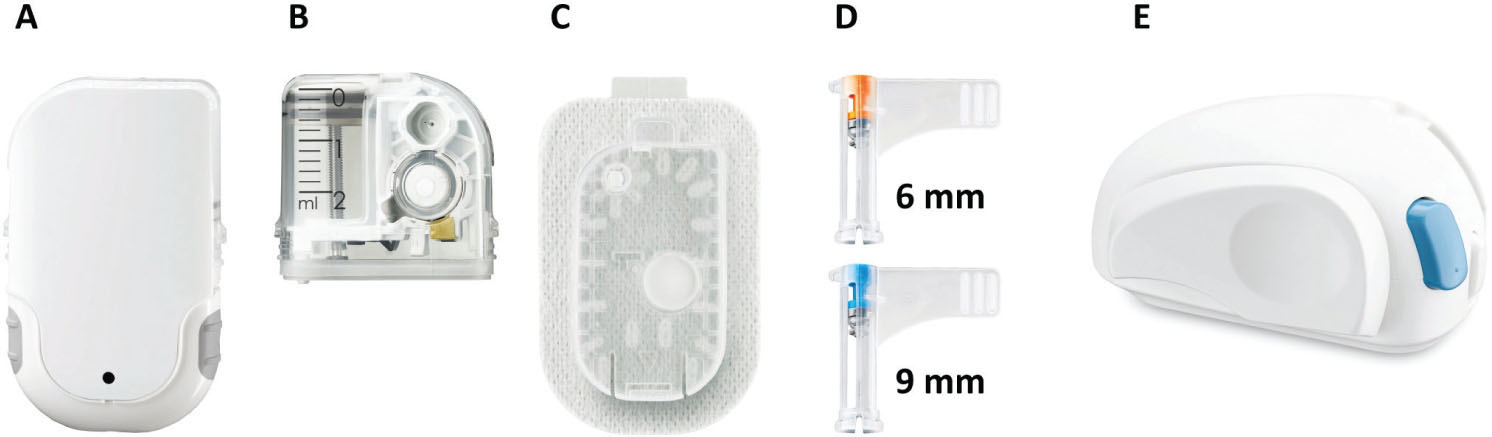
Fig. 2 - The parts that make up the pump: (A) pump base, (B) reservoir, (C) pump stand, (D) cannula with casing, (E) insertion device.
Moreover, this device is characterized by the peculiarity that its single components can be replaced independently, thus preventing the early production of any plastic waste, as it happens, instead, in non-modular systems (25).
With respect to this last point, during the development of these devices, due consideration was given to the importance of environmental protection, trying to keep the use of resources as well as the quantity of emissions as low as possible (36).
In this regard, a very recent study compared the Accu-Chek® Solo pump with two catheter-less pumps of non-modular design (Omnipod and Omnipod DASH) from a purely ecological standpoint. To this end, the three devices were disassembled and subjected to a series of measurements and evaluations; in particular, the following were determined for each system: the volumes of waste generated during a year of use, the recyclability of the materials, and, finally, the Global Warming Potential (GWP) (36).
In terms of the amount of waste at 1 year of use, using the best-case scenario where the individual components are used as per the manual, it was found that using the Accu-Chek® Solo pump results in 5.5 kg of waste compared to 4.9 kg for the Omnipod and 5.1 kg for the Omnipod DASH. However, the study showed that the modular design of Accu-Chek® Solo promotes longer periods of use of individual components and reduces the volume of product waste over a longer observation period (36).
When it comes to material recyclability, Accu-Chek® Solo has a recycling rate of 44.3% (2.44 kg) vs 14.6% (0.72 kg) for Omnipod and 16.0% (0.78 kg) for Omnipod DASH. In terms of GWP assessment, Accu-Chek® Solo was found to cause a lower level of CO2-equivalent emissions of 13.6 kg per year compared to 15.5 kg for non-modular systems (12% lower emissions). In conclusion, the examined study showed that a modular system of a micro-pump without a catheter can be considered more ecologically sound (36).
In addition, the Accu-Chek® Solo can be used by multiple patients because the reservoir, which is transparent to monitor residual insulin levels and detect air bubbles during the filling process, can accommodate different amounts of insulin (controlled filling up to 200 U, lasting up to 4 days). The reservoir can carry as little as 80 U of insulin, which is useful for patients who require small daily amounts of the hormone (25).
The design of the Diabetes Manager for remote control, similar to a smartphone, makes therapy management discreet yet intuitive, even when in public (25).
Thanks to the rechargeable Diabetes Manager device, bolus commands and basal rate settings can be sent directly to the pump, just as the pump can receive certain data from the diabetes manager (e.g., some reservoir level information or alarm alerts). If the patient cannot use the Diabetes Manager, insulin boluses can be administered directly and easily using the buttons on the sides of the pump (25).
The Diabetes Manager also has an integrated glucose meter (which requires the use of Accu-Chek Aviva/Performa test strips), which has the Accu-Chek bolus advisor feature. In addition, all the readings taken by the device can be shared with the patient’s diabetes team for analysis and interpretation by connecting the Diabetes Manager to a PC using Accu-Chek Smart Pix software (25).
An online platform, the Accu-Chek® Academy, is available for training and learning, a virtual space that can help patients quickly find the answers they need (25).
While all of these features address the specific needs of patients with T1DM, Accu-Chek® Solo provides improved control of blood glucose levels, reduced glycemic variability, and a positive impact on QoL. This is what emerged from the analysis of the three clinical cases previously discussed. Although we are aware of the methodological limitations of the following analysis and of the need to consider a larger number of cases in the near future, we were able to find a unified trend regarding the impact of the system on HbA1c, glycemic variability, TIR, TAR, and TBR, after about 4 months of use.
In particular, the three clinical cases analyzed in aggregate showed a reduction in HbA1c values and glycemic variability (mean blood glucose levels, SD and CV) and an improvement in TIR, with a consequent reduction in TAR and TBR (Tab. VI).
Of particular interest were also the data regarding the impact of CSII on QoL, as the analysis of the questionnaires showed positive results for the three clusters and in line with expectations (Tab. IV):
– Factor 1 “Benefits/Advantages”: agree-strongly agree-agree;
– Factor 2 “Social restrictions”: disagree-neutral-disagree;
– Factor 3 “Impact on the psychological sphere”: neutral-disagree-neutral.
| Baseline Delta – First Visit | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| HbA1c (%) | –0.7 | –0.8 | –0.7 |
| Mean blood glucose level (mg/dL) | –26 | –25 | –18 |
| Hypoglycemia episodes (n) | 0 | ||
| TIR 70-180 mg/dL (%) | 24.0 | 28.9 | 12.0 |
| TAR >180 mg/dL (%) | –19.0 | –9.2 | –12.0 |
| TBR <70 mg/dL (%) | –5.0 | 0.3 | 0.0 |
| SD (mg/dL) | –36.5 | –6.0 | |
| CV (%) | 14.6 | –19.2 | –24.0 |
| CGM usage time (%) | 21.0 | 3.0 | 0.0 |
CGM = continuous glucose monitoring; CSII = continuous subcutaneous insulin infusion; CV = coefficient of variation; HbA1c = glycated hemoglobin; SD = standard deviation; T1DM = type 1 diabetes mellitus; TAR = time above range; TBR = time below range; TIR = time in range.
Equally positive results emerged from the second questionnaire, administered to ascertain the level of satisfaction of the users regarding the use of the pump (Tab. V).
The three patients were particularly “very satisfied” with the absence of a catheter and the possibility of delivering the bolus directly from the pump. The procedures for placing the device are considered simple and, among the most appreciated features are the convenience of the temporal variation of the bolus, the absence of a catheter, and that the device is small and discreet.
Finally, patients reported that they were generally “satisfied/very satisfied” with the system and were unanimous in their intention to continue using Accu-Chek® Solo.
In conclusion, it is desirable that more and more consideration be given to the different reasons behind the reduced diffusion of CSII in Italy, in order to reverse this trend.
Given the complexity of the framework analyzed, it will be necessary to undertake a series of complementary strategies that can take into account the many facets of psychological and social nature involved.
The multidisciplinary team, expert in CSII, will be called in first person to take charge of carefully selecting and guiding the patient step by step, during the transition to CSII therapy and in maintaining therapeutic adherence, both from an educational point of view as well as psychological/motivational support.
At the same time, a new generation of catheter-less insulin pumps, the Accu-Chek® Solo, will be available, which, in addition to the system’s ease of use and patient-friendly design, would provide a number of clinical management benefits.
From the analysis and overcoming of the limiting factors discussed so far, it is likely that we will see a gradual decrease in barriers to the use of CSII therapy and a reduction in drop-out among patients with T1DM on insulin pump therapy.
Acknowledgements
Editorial support was provided by Health Publishing & Services Srl funded by Roche Diabetes Care, Roche Italia. Click here to access a portal dedicated to Healthcare Professional with the latest information, medical education resources, and customized services on diabetes.
Disclosures
Conflict of interest: KE reports personal fees, outside the submitted work, from: Abbott, Lifescan, Theras, Novo Nordisk, Astrazeneca, Sanofi, Mundipharma, Eli Lilly, and Bruno. AMB reports personal fees from Roche Diabetes Care, outside the submitted work. FC has nothing to disclose. MD has nothing to disclose. CM reports personal fees from Roche Diabetes Care Italy, non-financial support from Eli Lilly, non-financial support from Movi, outside the submitted work. CN has nothing to disclose. MP reports personal fees from Roche, from Abbott and Lifescan, outside the submitted work. EB reports personal fees from Abbott Diabetes Care, from Medtronic, from Roche Diabetes Care, outside the submitted work.
Financial support: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- 1. Associazione Medici Diabetologi (AMD) – Società Italiana di Diabetologia (SID). Standard italiani per la cura del diabete mellito. 2018. Online (Accessed March 17, 2021).
- 2. Addendum. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes – 2020. Diabetes Care. 2020;43(suppl 1):S98-S110 – August 01, 2020. CrossRef PubMed
- 3. Commissariat PV, Boyle CT, Miller KM, et al. Insulin pump use in young children with type 1 diabetes: sociodemographic factors and parent-reported barriers. Diabetes Technol Ther. 2017;19(6):363-369. CrossRef PubMed
- 4. Jeitler K, Horvath K, Berghold A, et al. Continuous subcutaneous insulin infusion versus multiple daily insulin injections in patients with diabetes mellitus: systematic review and meta-analysis. Diabetologia. 2008;51(6):941-951. CrossRef PubMed
- 5. Blackman SM, Raghinaru D, Adi S, et al. Insulin pump use in young children in the T1D Exchange clinic registry is associated with lower hemoglobin A1c levels than injection therapy. Pediatr Diabetes. 2014;15(8):564-572. CrossRef PubMed
- 6. Johnson SR, Cooper MN, Jones TW, Davis EA. Long-term outcome of insulin pump therapy in children with type 1 diabetes assessed in a large population-based case-control study. Diabetologia. 2013;56(11):2392-2400. CrossRef PubMed
- 7. Pozzilli P, Battelino T, Danne T, Hovorka R, Jarosz-Chobot P, Renard E. Continuous subcutaneous insulin infusion in diabetes: patient populations, safety, efficacy, and pharmacoeconomics. Diabetes Metab Res Rev. 2016;32(1):21-39. CrossRef PubMed
- 8. Documento del Gruppo di Studio Intersocietario AMD-SID-SIEDP “Tecnologia e Diabete”. 2019. Online (Accessed March 17, 2021).
- 9. American Diabetes Association. Continuous subcutaneous insulin infusion. Diabetes Care. 2004;27(suppl 1):S110. CrossRef PubMed
- 10. Bonora E, Sesti G. Società Italiana di Diabetologia. Il diabete in Italia. Bononia University Press; 2016. Online (Accessed March 17, 2021).
- 11. Manicardi V, Adinolfi V, Aricò N, et al. Annali AMD 2020. Diabete di tipo 1. 2020. Online (Accessed March 17, 2021).
- 12. Renard E. Insulin pump use in Europe. Diabetes Technol Ther. 2010;12(S1)(suppl 1):S29-S32. CrossRef PubMed
- 13. Tanenbaum ML, Hanes SJ, Miller KM, Naranjo D, Bensen R, Hood KK. Diabetes device use in adults with type 1 diabetes: barriers to uptake and potential intervention targets. Diabetes Care. 2017;40(2):181-187. CrossRef PubMed
- 14. Ronsin O, Jannot-Lamotte MF, Vague P, Lassman-Vague V. Factors related to CSII compliance. Diabetes Metab. 2005;31(1):90-95. CrossRef PubMed
- 15. Cortina S, Repaske DR, Hood KK. Sociodemographic and psychosocial factors associated with continuous subcutaneous insulin infusion in adolescents with type 1 diabetes. Pediatr Diabetes. 2010;11(5):337-344. CrossRef PubMed
- 16. de Vries L, Grushka Y, Lebenthal Y, Shalitin S, Phillip M. Factors associated with increased risk of insulin pump discontinuation in pediatric patients with type 1 diabetes. Pediatr Diabetes. 2011;12(5):506-512. CrossRef PubMed
- 17. Tanenbaum ML, Adams RN, Hanes SJ, et al. Optimal use of diabetes devices: clinician perspectives on barriers and adherence to device use. J Diabetes Sci Technol. 2017;11(3):484-492. CrossRef PubMed
- 18. Seereiner S, Neeser K, Weber C, et al. Attitudes towards insulin pump therapy among adolescents and young people. Diabetes Technol Ther. 2010;12(1):89-94. CrossRef PubMed
- 19. Sullivan-Bolyai S, Knafl K, Tamborlane W, Grey M. Parents’ reflections on managing their children’s diabetes with insulin pumps. J Nurs Scholarsh. 2004;36(4):316-323. CrossRef PubMed
- 20. Lombardo F, Scaramuzza AE, Iafusco D. Failure of glycated hemoglobin drop after continuous subcutaneous insulin infusion initiation may indicate patients who discontinue: a 4-year follow-up study in children and adolescents with type 1 diabetes. Acta Diabetol. 2012;49(S1)(suppl 1):S99-S105. CrossRef PubMed
- 21. Wong JC, Boyle C, DiMeglio LA, et al; T1D Exchange Clinic Network. Evaluation of pump discontinuation and associated factors in the T1D Exchange Clinic Registry. J Diabetes Sci Technol. 2017;11(2):224-232. CrossRef PubMed
- 22. Hofer SE, Heidtmann B, Raile K, et al; DPV-Science-Initiative and the German working group for insulin pump treatment in pediatric patients. Discontinuation of insulin pump treatment in children, adolescents, and young adults. A multicenter analysis based on the DPV database in Germany and Austria. Pediatr Diabetes. 2010;11(2):116-121. CrossRef PubMed
- 23. Wong JC, Dolan LM, Yang TT, Hood KK. Insulin pump use and glycemic control in adolescents with type 1 diabetes: predictors of change in method of insulin delivery across two years. Pediatr Diabetes. 2015;16(8):592-599. CrossRef PubMed
- 24. Wood JR, Moreland EC, Volkening LK, Svoren BM, Butler DA, Laffel LM. Durability of insulin pump use in pediatric patients with type 1 diabetes. Diabetes Care. 2006;29(11):2355-2360. CrossRef PubMed
- 25. Ulbrich S, Waldenmaier D, Haug C, Freckmann G, Rendschmidt T, Künsting T. Concept and implementation of a novel patch pump for insulin delivery. J Diabetes Sci Technol. 2020;14(2):324-327. CrossRef PubMed
- 26. Heinemann L, Waldenmaier D, Kulzer B, Ziegler R, Ginsberg B, Freckmann G. Patch pumps: are they all the same? J Diabetes Sci Technol. 2019;13(1):34-40. CrossRef PubMed
- 27. Layne JE, Parkin CG, Zisser H. Efficacy of a tubeless patch pump in patients with type 2 diabetes previously treated with multiple daily injections. J Diabetes Sci Technol. 2017;11(1):178-179. CrossRef PubMed
- 28. Layne JE, Parkin CG, Zisser H. Efficacy of the OmniPod insulin management system on glycemic control in patients with type 1 diabetes previously treated with multiple daily injections or continuous subcutaneous insulin infusion. J Diabetes Sci Technol. 2016;10(5):1130-1135. CrossRef PubMed
- 29. Danne T, Schwandt A, Biester T, et al; DPV Initiative. Long-term study of tubeless insulin pump therapy compared to multiple daily injections in youth with type 1 diabetes: data from the German/Austrian DPV registry. Pediatr Diabetes. 2018;19(5):979-984. CrossRef PubMed
- 30. Mehta SN, Tinsley LJ, Kruger D, et al. Improved glycemic control following transition to tubeless insulin pump therapy in adults with type 1 diabetes. Clin Diabetes. 2021;39(1):72-79. CrossRef PubMed
- 31. Mader JK, Oliver N, Vesper I, et al. First results from PRO Solo: patient reported outcomes from a clinical trial comparing a new patch pump with MDI and an established patch pump. EASD, Abstract # 708. Online (Bergh). 2020. Accessed May 9, 2021.
- 32. Sakane N, Murata T, Tone A, et al. Development and validation of the Continuous Subcutaneous Insulin Infusion-Related Quality-of-Life (CSII-QOL) scale. Diabetes Technol Ther. 2020;22(3):216-221. CrossRef PubMed
- 33. SID. Indagine della società italiana di diabetologia (SID) sulle differenze regionali nell’accesso a farmaci, presidi e servizi per la persona con diabete. Online (Bergh). 2019. Accessed March 17, 2021.
- 34. Roze S, Duteil E, De Brouwer BF, de Portu S. Health-economic analysis of the use of Sensor-Augmented Pump (SAP) therapy in the Netherlands compared to insulin pump therapy alone (CSII), in type 1 diabetic patients. Value Health. 2015;18(7):A360-A361. CrossRef
- 35. Ziegler R, Oliver N, Waldenmaier D, Mende J, Haug C, Freckmann G. Evaluation of the accuracy of current tubeless pumps for continuous subcutaneous insulin infusion. Diabetes Technol Ther. 2020;dia.2020.0525. CrossRef PubMed
- 36. Winkelkötter J, Reitz T. Analysis of the waste volume and CO2-equivalent caused by the use of selected patch pumps. J Diabetes Sci Technol. 2021. CrossRef PubMed